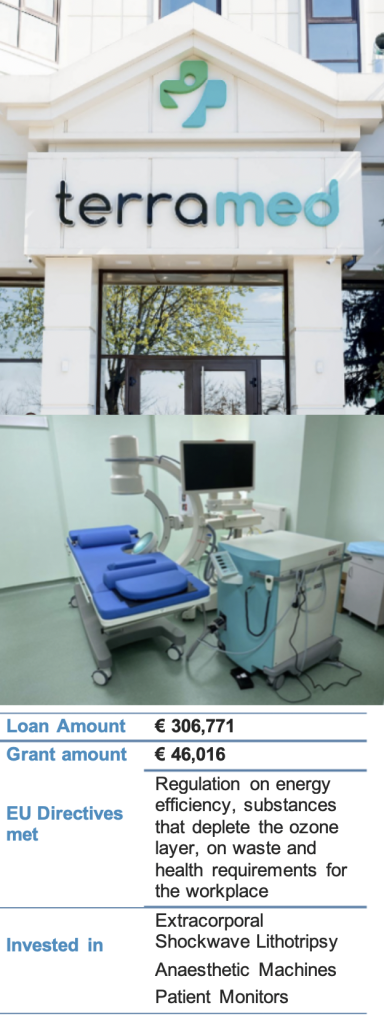
The company MedPharmConsult SRL has been providing medical services at the two sites of its Terramed clinic since 2011. The clinics have always provided high-level medical services with highly trained staff and modern equipment, offering a comprehensive range of medical and dental treatments for adults as well as children.
The company upgraded key equipment in one of the clinics to increase the intervention precision as well as to improve its occupational health and safety standards. The investment allows the company to offer medical services and working conditions compliant with local as well as EU standards. After the successful project verification the company received 15% of the loan value as a grant incentive, funded under the EU4Business initiative of the European Union.
With the investment, the company now meets a wide variety of European standards, including:
- Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017 on medical devices, amending Directive 2001/83/EC, Regulation (EC) No 178/2002 and Regulation (EC) No 1223/2009 and repealing Council Directives 90/385/EEC and 93/42/EEC
- Directive 2013/35/EU of The European Parliament and of the Council on the minimum health and safety requirements regarding the exposure of workers to the risks arising from physical agents (electromagnetic fields) (20th individual Directive within the meaning of Article 16(1) of Directive 89/391/EEC) and repealing Directive 2004/40/EC
- Directive 2013/59/Euratom – protection against ionising radiation of 5 December 2013 laying down basic safety standards for protection against the dangers arising from exposure to ionising radiation, and repealing Directives 89/618/Euratom, 90/641/Euratom, 96/29/Euratom, 97/43/Euratom and 2003/122/Euratom
- Directive 2014/35/EU of the European Parliament and of The Council of 26 February 2014 on the harmonisation of the laws of the Member States relating to the making available on the market of electrical equipment designed for use within certain voltage limits
- Directive 2006/42/EC of the European Parliament and of The Council of 17 May 2006 on machinery, and amending Directive 95/16/EC (recast)
- Directive 2014/30/EU of the European Parliament and of The Council of 26 February 2014 on the harmonization of the laws of the Member States relating to electromagnetic compatibility (recast)
Directive 2014/68/EU on the harmonisation of the laws of the Member States relating to the making available on the market of pressure equipment (recast)